Uncovering the Split ISR Pathway
A New Understanding of Cellular Stress and Its Implications for Treating Genetic Diseases
A recent study has challenged the long-standing understanding of how our cells respond to stress. For decades, scientists believed that cells react to various stressors—such as starvation, infection by pathogens, heat, and iron deficiency—through a unified pathway known as the Integrated Stress Response (ISR). The ISR functions by modulating protein synthesis, conserving energy, and directing resources where they are most needed. This process decreases the production of housekeeping proteins while increasing the synthesis of stress-response proteins, allowing the body to adapt to challenging conditions.
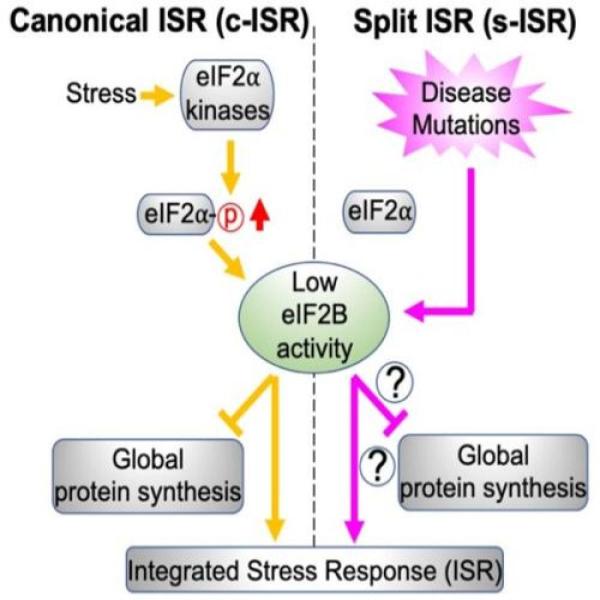
Traditionally, these stressors have been considered external, but authors of a new study questioned whether the ISR truly applies universally to all forms of stress, both external and internal. A recent paper, “Plasticity of the mammalian integrated response” published in Nature, reveals that when cells face internal stressors—such as genetic mutations—the stress response follows an alternative pathway, which they have termed the "split ISR" (sISR).
At the heart of the ISR pathway is the ATF4 protein, which plays a key role in regulating genes related to metabolism and facilitates a coordinated stress response. Typically, the activation of ATF4 is triggered by the phosphorylation of a multi-protein factor called eIF2α that plays a central role in initiating protein synthesis. However, the researchers discovered that in the sISR pathway, ATF4 activation still occurs, but not through eIF2α phosphorylation. Instead, it is triggered by a different protein. This distinct mechanism results in a unique rewiring of cellular metabolism that helps restore cellular homeostasis.
Beyond challenging the idea that the ISR is the universal pathway for responding to stress, regardless of the stressor’s nature or duration, the discovery of sISR has significant implications for the study of genetic diseases. Many genetic disorders involve chronic internal stress, and understanding this alternative stress response could open new avenues for targeted therapies. This insight may lead to promising opportunities for treating genetic conditions that continuously stress cells.
Vanishing White Matter Disease (VWMD), a brain disorder caused by mutations in eIF2B, is just one example of a genetic disease caused by persistent "internal stress." In VWMD, mutations weaken cellular stress response systems, making brain cells especially vulnerable to normal physiological pressures. This concept applies to many other genetic disorders, where inherited mutations impair a cell’s ability to manage internal stress, leading to progressive damage and disease.
Dr. Jonathan Dinman, Director, Institute for Bioscience and Biotechnology Research (IBBR) and Professor in the Department of Cell Biology and Molecular Genetics at the University of Maryland, College Park is a co-author on the paper and stated, “Understanding VWMD helps illustrate how chronic cellular stress contributes to a range of genetic conditions. As such, it can be used as a paradigm for understanding how genetic mutations cause human disease through the sISR, and from there, how to develop effective therapeutic strategies for a wide range of patients.
Originally published by the Institute for Bioscience and Biotechnology Research (IBBR)






